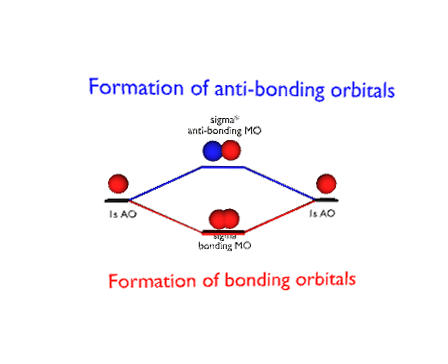
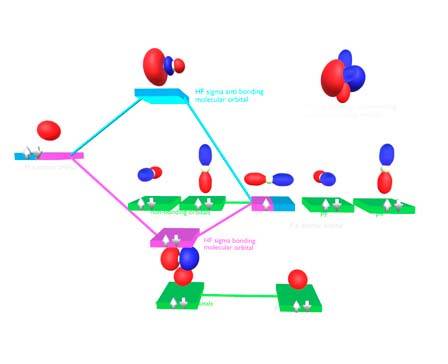
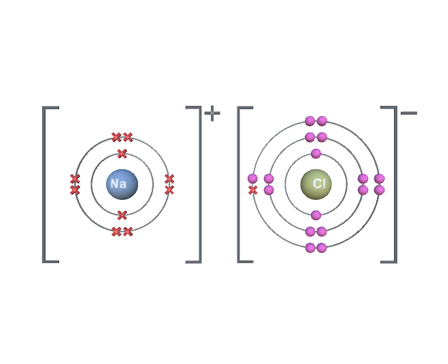
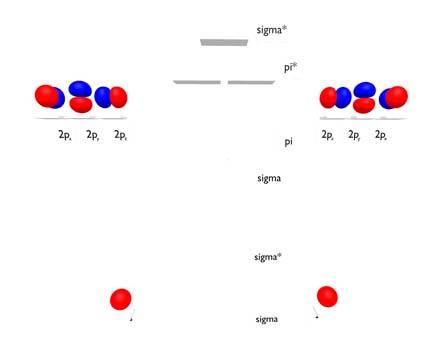
rare
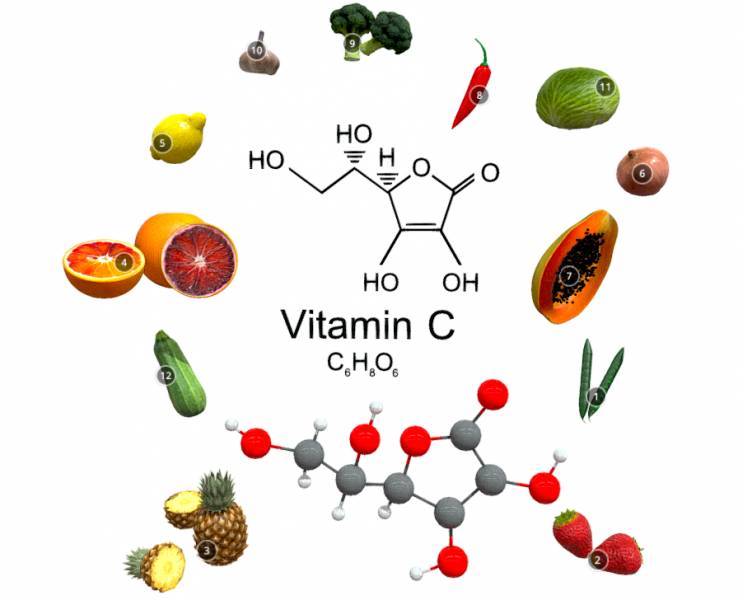
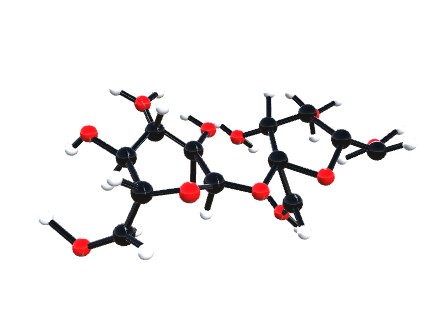
Vitamin C
Vitamin C, also known as ascorbic acid, has several important functions.
These include:
helping to protect cells and keeping them healthy maintaining healthy skin, blood vessels, bones and cartilage helping with wound healing Vitamin C is a water-soluble vitamin. This means that it dissolves in water and is delivered to the body’s tissues but is not well stored, so it must be taken daily through food or supplements.
Fruits and vegetables are the best sources of this vitamin: citrus, bell peppers, strawberries, tomatoes, cruciferous vegetables (broccoli, Brussels sprouts, cabbage, cauliflower), etc.
Apri esperienza