Public experience
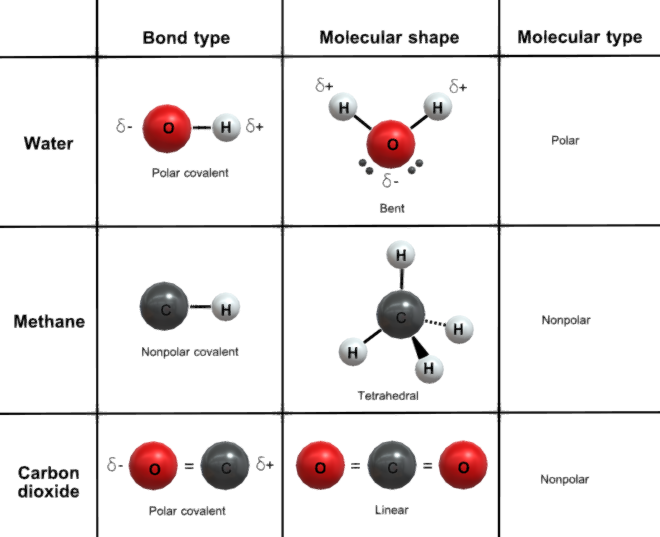
Polar and nonpolar covalent bonds
Polar covalent bonds involve unequal sharing of electrons between atoms due to different electronegativities, creating partial charges. Example: water (H₂O). Nonpolar covalent bonds involve equal sharing of electrons between atoms with similar electronegativities, resulting in no charge separation. Examples: nitrogen (N₂), methane (CH₄).
Related library categories
Access
ARLOOPA app
Categories
1
Published
Dec 10, 2024
Scene assets
1
Open on mobile
Scan the QR code to launch the experience
Use your phone camera to open the correct destination for this experience. WebAR experiences open in the browser. App-based experiences open through the ARLOOPA experience flow.
ARLOOPA app launch QR
Scan this code on your phone
How it works
How people usually open this experience
ARLOOPA Studio
Create your own AR experience
Use ARLOOPA Studio to build and publish a similar experience for your campaign, venue, product, or audience.





