Public experience
The polarity of water
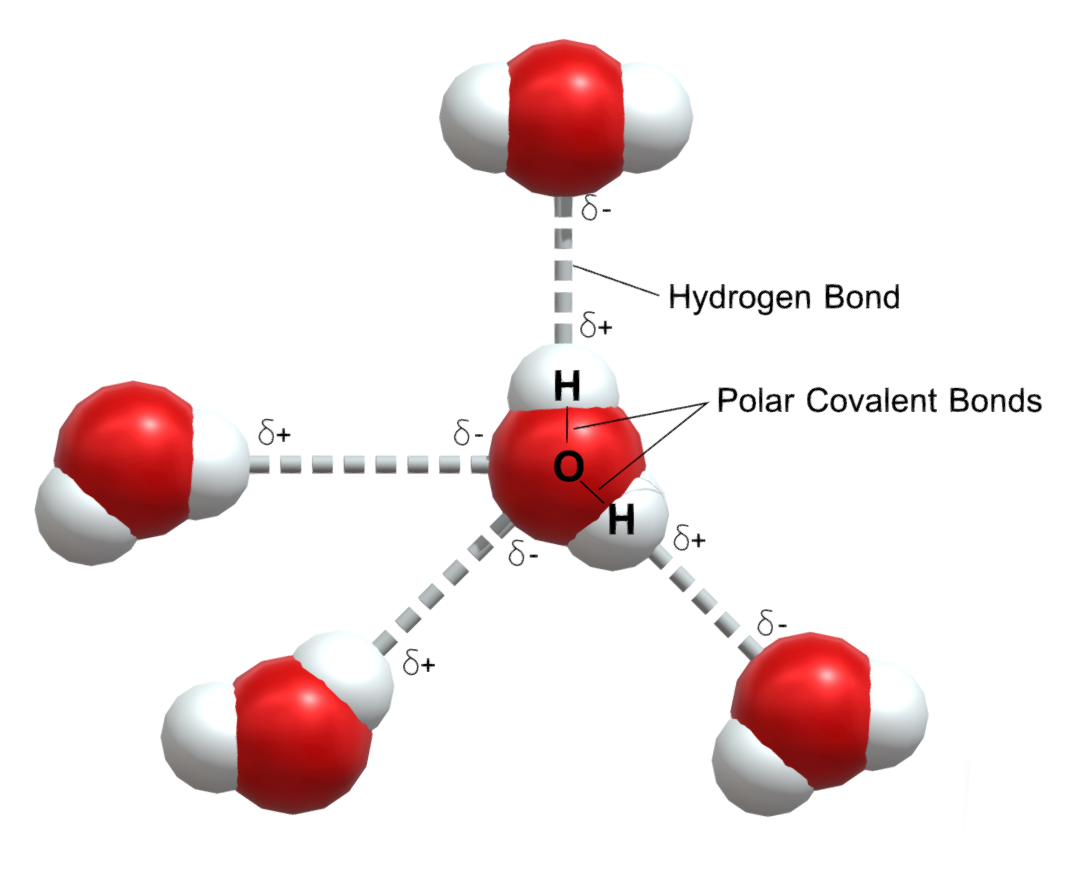
Water (H₂O) is a polar molecule because it has a bent shape and an unequal distribution of electrons. Oxygen is more electronegative than hydrogen, causing the electrons to be more concentrated around the oxygen atom. This results in a partial negative charge on the oxygen and partial positive charges on the hydrogen atoms, creating a dipole moment. This polarity allows water to form hydrogen bonds and dissolve many substances, making it a versatile solvent.
Related library categories
Access
ARLOOPA app
Categories
1
Published
Dec 10, 2024
Scene assets
1
Open on mobile
Scan the QR code to launch the experience
Use your phone camera to open the correct destination for this experience. WebAR experiences open in the browser. App-based experiences open through the ARLOOPA experience flow.
ARLOOPA app launch QR
Scan this code on your phone
How it works
How people usually open this experience
ARLOOPA Studio
Create your own AR experience
Use ARLOOPA Studio to build and publish a similar experience for your campaign, venue, product, or audience.





